Consider diffusion at the front and rear surfaces of an incremental planar volume. Fick's 2nd law of diffusion describes the rate of accumulation (or depletion) of concentration within the volume as proportional to the local curvature of the concentration gradient. The local rule for accumulation is given by Fick's 2nd law of diffusion:
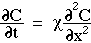
in which the accumulation, dC/dt [cm-3 s-1], is proportional to the diffusivity 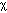 [cm2/s] and the 2nd derivative (or curvature) of the concentration,
[cm2/s] and the 2nd derivative (or curvature) of the concentration, 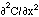 [cm-3 cm-2] or [cm-5]. The accumulation is positive when the curvature is positive, i.e., when the concentration gradient is more negative on the front end of the planar volume and less negative on the rear end so that more flux is driven into the volume at the front end than is driven out of the volume at the rear end.
[cm-3 cm-2] or [cm-5]. The accumulation is positive when the curvature is positive, i.e., when the concentration gradient is more negative on the front end of the planar volume and less negative on the rear end so that more flux is driven into the volume at the front end than is driven out of the volume at the rear end.
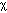 [cm2/s] and the 2nd derivative (or curvature) of the concentration,
[cm2/s] and the 2nd derivative (or curvature) of the concentration, 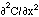 [cm-3 cm-2] or [cm-5]. The accumulation is positive when the curvature is positive, i.e., when the concentration gradient is more negative on the front end of the planar volume and less negative on the rear end so that more flux is driven into the volume at the front end than is driven out of the volume at the rear end.
[cm-3 cm-2] or [cm-5]. The accumulation is positive when the curvature is positive, i.e., when the concentration gradient is more negative on the front end of the planar volume and less negative on the rear end so that more flux is driven into the volume at the front end than is driven out of the volume at the rear end. 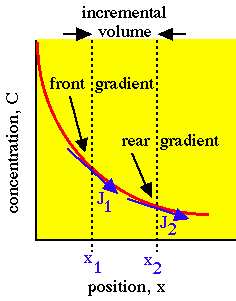
Incremental planar volume accumulates concentration because the front gradient at x1 drives more flux J1 into the volume than the flux J2 driven out of the volume by the rear gradient at x2.
Agustin Egui
CAF
The differential equation for optical diffusion is simply Fick's 2nd law with the substitution of the product cD for the diffusivity 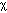 and substitution of F/c for concentration C, although the 1/c factors introduced on both sides of the equation cancel:
and substitution of F/c for concentration C, although the 1/c factors introduced on both sides of the equation cancel:
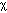 and substitution of F/c for concentration C, although the 1/c factors introduced on both sides of the equation cancel:
and substitution of F/c for concentration C, although the 1/c factors introduced on both sides of the equation cancel: 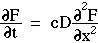
No hay comentarios:
Publicar un comentario